
Universal Constants
In 1900, Max Plank made a major discovery, one which ranked with the discoveries of Newton. The philosophy and the paradigm of physics, after his discovery never were to be the same, although it took another 27 years for quantum mechanics to fall. His discovery dealt with the fundamental units of matter and energy. It provides equations that define with great accuracy the laws governing the propagation of radiant energy. The extraordinary feature of this theory was that it rested on the assumption that radiant energy is emitted not in an unbroken stream but in discontinuous packets which Planck termed 'quanta'.
Planck is the discoverer of the Planck's Constant1 which is a number which never changes. It is used to calculate the size of the quanta of each light-frequency [colour]. On purely theoretical grounds he concluded that each quantum (packet) carried an amount of energy given by the equation, E=hv where v is the frequency of radiation and h is Planck's constant. A small but inexorable number, which has since proved to be one of the most fundamental constants in nature. All forms of radiant energy - light, heat, X-rays - actually travel through space in separate and discontinuous quanta and the amount of emitted energy divided by the frequency is always equal to h.
Thus sensation of colour arise from the bombardment of our optic nerves by light quanta which differ from each other just as the frequency varies in the equation E=hv. Thus, energy-packets of each colour would have the same amount of energy. The packets of violet light, however, are larger than the packets of green light and these are again larger than packets of red light.
Another universal constant is the velocity of light, denoted by the letter c, which was accurately determined in 1849 to be 186,284 miles per second2. The velocity of light is unaffected by the motion of earth, sun, moon, star or any other system moving anywhere in the universe i.e. it is constant throughout the universe and is unaffected either by the motion of its source or the motion of the receiver. We are already familiar with Einstein's equation giving the increase in mass3 with velocity of a moving body, where this constant is used -
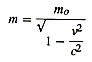
when m0 is mass of a stationary object or rest mass, m is its mass when in motion, v is its velocity, c is the velocity of light.
From the above equation, we can see that as v increases the mass also increases and when v is equal to c, mass will be infinity We had come across an instance of increase in mass while discussing the characteristics of subatomic particles. From this it is clear that the speed of a particle of matter is always less than that of light and other radiations.
1 The latest value of Planck's constant is determined to be 6.62517 x joule-seconds.
2 The latest corrected value is determined to be 2.997925 x 108 meters per second in vacuum
2 Units of measurement:
There are three fundamental units of modern scientific measurement. They are units of length, mass and time. The unit of length, metre, was defined earlier as one ten-millionth part of a quarter meridian. In 1967, this was replaced by the wavelength of orange-red radiation of krypton86. The present valid definition of the unit of length is "1,650,763,73 wave-lengths of orange-red radiation of krypton86 in vacuum"
Initially the unit of mass ‘gramme’ was considered the mass of a cubic centimetre of water at its maximum density. Later on, in MIS system, this was changed to kilogram i.e. the mass of a cubic decimeter of water. But this suffered from the inherent uncertainty associated with the measurement of volume and temperature. In 1960, kilogram was independently defined as the mass of a platinumiridium cylinder which is preserved at International Bureau of Weights & Measures at Serves, Paris.
This definition of mass has been considered in a mode rather than a rule. So the defining of both mass and length in terms of a certain wavelength of light may provide a coherent means. The wave and particle nature of light inspired S. K. Mukherjee and A. Choudhury to conceive the mass-equivalence of the particle associated with the orange-red radiation of krypton86 and a suggestion was put forward by them to define kilogram in terms of the mass of a particle whose De Broglie wave- length is already
taken for standardisation of length. Thus the kilogram can be considered in terms of the number of particles associated with the radiation in order to follow the international convention standards. A precise estimation of the number of particles assumes the value 2.74118798 x 10 35We shall discuss the unit of time in the following soon.
 Jethalal S. Zaveri
Jethalal S. Zaveri
 Prof. Muni Mahendra Kumar
Prof. Muni Mahendra Kumar

